
The best way to mark the oblique ones is tracing these lines with a pencil from behind, placing the printed figure upside down in a lighted window. Mark (unpainted) vertical and oblique lines (the ends of which are indicated by dashed red lines on the template). Print the template seen in figure 2 on thick paper. (You will find both Word and PDF documents of the figure in the Supporting Information below.)įigure 2: The template of the Kaleidocycle. It is a playful study tool that belongs to each student. This device is mainly destined for students in high school who are studying chemistry. It is intended that students make their own figure from the template with the help of the teacher according to the instructions that we will develop in detail below. Each student can use it not only in class but also at home with friends or family members. Video 1: Kaleidocycle of the Periodic Table Who is the kaleidocycle for? On these, four different pictures can be displayed. In this three-dimensional figure of the periodic table are the elements organized in four blocks according to their final electronic structure. It is intended that students with this playful figure actively participate in classes by rotating their kaleidocycle looking for the groups or elements that are being studied. The entire periodic table fits in one palm of their hands. It is also a didactic device because students only focus their attention on one block or group of elements from the entire Periodic Table. It can be a more entertaining, motivating and exciting way of learning about the subject of the Periodic Table. See figure 1. By turning it you can easily choose one of the four faces. As high school teachers, we decided to make a figure of the Periodic Table that would be attractive and catching for our chemistry students. For this purpose, we came up with the idea of using a kaleidocycle, which is a three-dimensional figure obtained by folding a flat template.Ī kaleidocycle has four different faces, each one made of a juxtaposition of rhombuses. The groups will then present their findings to the class and discuss how they arrived at their findings.Īs a class we will group the elements under the three groups after discussing the criteria for assigning elements to groups.In 2019, the 150th anniversary of the creation of the first Periodic Table established by Dimitri Mendeleiev was commemorated. They will arrange the samples into metals, nonmetals and metalloids. They will discuss their findings in their own small groups and group the samples under two categories based on their physical and chemical properties and find which elements could fit into either of the groups and why. The student groups will then make a data table to compare the observations for the given samples. With copper (II) chloride which is a blue colored solution, they will be looking for color change of the liquid and of the sample. 
To each sample, 15 to 20 drops of the acid solution will be added and they will look for any reaction (change ) happening like bubbles of hydrogen gas coming out. 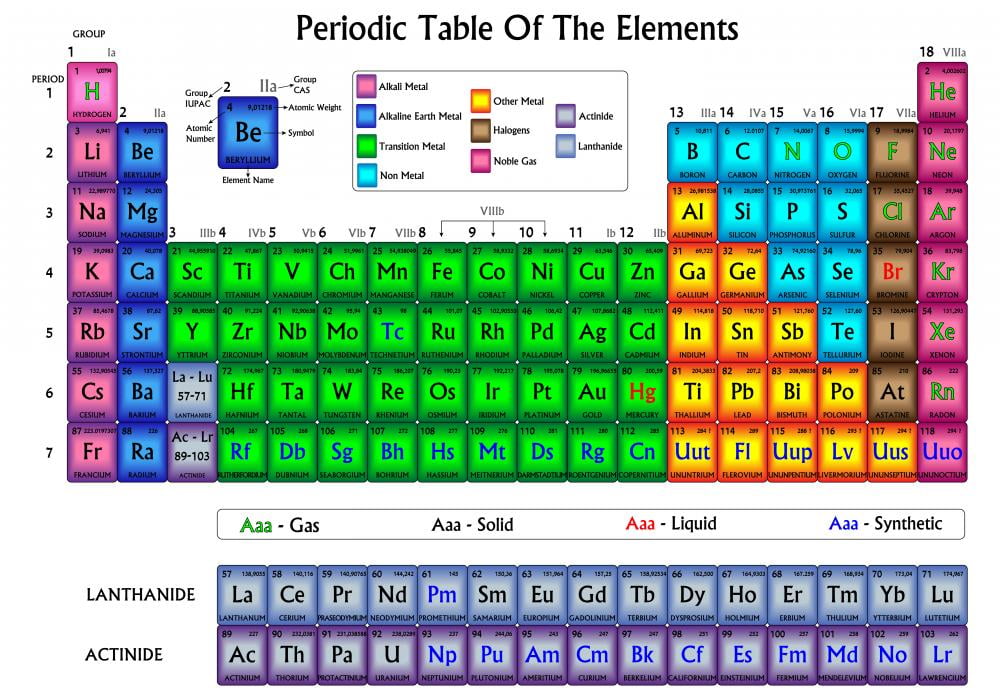
They will take a small portion of the given samples in a well plate. Then they will test for the reactivity of the samples with 0.5M hydrochloric acid and 0.1M copper (II) chloride solution. They will use the conductivity tester to see if the sample conducts electricity. They will use the hammer to test if the sample is brittle or malleable. They will look at the appearance of the given sample to see if it has luster or is dull. The students have to group the samples under the categories of metals, nonmetals or metalloids. The samples the student groups get are carbon (pencil lead works), pieces of magnesium ribbon, silicon lumps, sulfur lumps, iron filings, and pieces of mossy zinc. /PeriodicTableallcolor-58b5c82c3df78cdcd8bbb80f.png)
The students will form small lab groups of two or three members.Īfter I review the difference between physical and chemical properties, and the characteristics of metals, nonmetals and metalloids, the student groups will get samples in seven vials which have been coded with letters 'a' to 'g'.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
